Our expertise in the field of human milk research, as well as our research into the complexities of human milk, has led to a number of pioneering discoveries that have redefined the world of infant formula.
Taking our inspiration from the unique composition of human milk, we have developed nutritional solutions that can modulate the gut microbiota in order to elicit positive health outcomes for infants and young children. Examples include our patented prebiotic scGOS/lcFOS mixture as well as our innovative Synbiotic blend that have shown clinically significant benefits for GI and immune health.
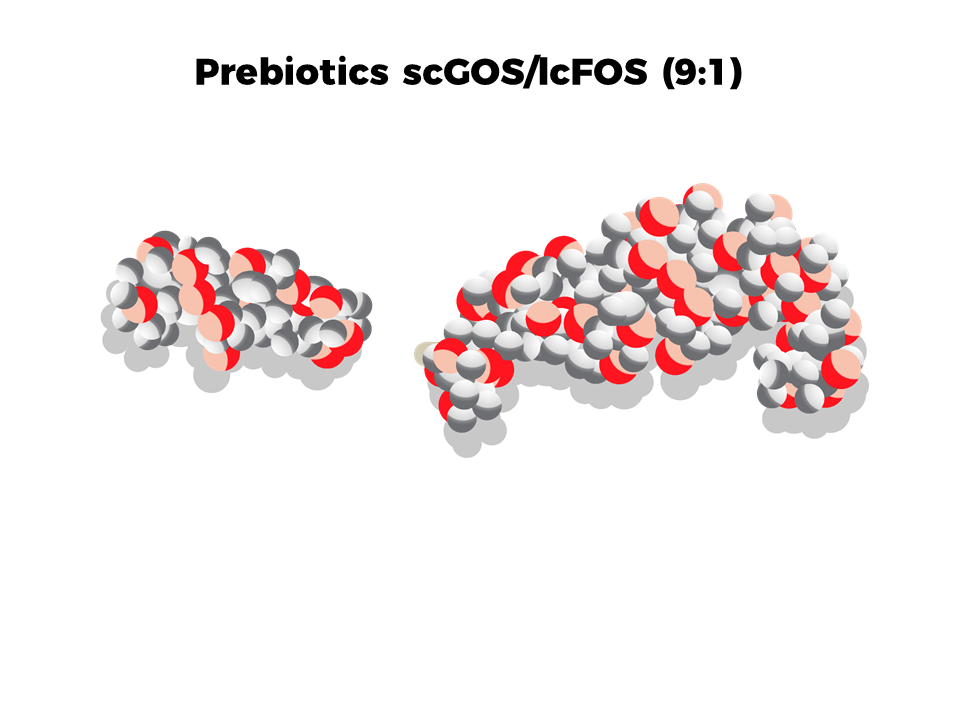
scGOS/lcFOS (9:1): our unique, patented prebiotic mixture
We were the first company to include prebiotic oligosaccharides in our infant formulas.
Introduced across our ranges in 2000, our unique, patented blend contains short chain galacto-oligosaccharides (scGOS) and long chain fructo-oligosaccharides (lcFOS), in a 9:1 ratio. Following almost 20 years of research, with a total of over 30 studies, described in >55 scientific publications, clinically proven health benefits include:
• a healthy microbiota, closer to breastfed infants1Moro, et al. J Pediatr Gastroenterol Nutr. Published on 2002; 34:291- 5 ,2Boehm, et al. Arch Dis Child. Published on 2002; 86: F178-81 ,3Knol, et al. J Pediatr Gastroenterol Nutr. Published on 2005a; 40:36-42 ,4Haarman, et al. Appl Environ Microbiol. Published on 2005; 71:2318-24 ,5Moro, et al. J Pediatr Gastroenterol Nutr. Published on 2002; 34:291-5 ,6Knol, et al. J Pediatr Gastroenterol Nutr. Published on 2002; 34:291-5 ,7Desci, et al. Orv Hetil. Published on 2005; 146:2445-50
• stool patterns closer to that of breastfed infants8Moro, et al. J Pediatr Gastroenterol Nutr. Published on 2002; 34:291- 5
• reduction in the incidence of gastroenteritis, eczema and allergy9Boehm, et al. Arch Dis Child. Published on 2002; 86: F178-81 ,10Bruzzese, et al. Clin Nutr. Published on 2009;28:156-161 ,11Arslanoglu, et al. J Nutr. Published on 2007; 137:2420-2424 ,12Arslanoglu, et al. J Nutr. Published on 2008; 138:1091-1095 ,13Arslanoglu, et al. J Biol Regul Homeost Agents. Published on 2012;26:59- 59
Synbiotics: the latest tool in the fight against allergy
Bifidobacterium breve is the most commonly isolated bifidobacterial species from human milk, and is found in the gut of breastfed infants.
Our studies have confirmed that Bifidobacterium breve M-16V is also the most promising strain in reducing allergic symptoms and in producing synergistic benefits when combined with our unique prebiotic mixture scGOS/lcFOS (9:1)
• Improves allergic symptoms in infants with IgE-mediated atopic dermatitis14van der Aa, L.B, et al. Clin Exp Allergy. Published on 2010. 40(5): p. 795-804
• Improves diaper dermatitis and GI symptoms15van der Aa, L.B, et al. Clin Exp Allergy. Published on 2010. 40(5): p. 795-804
• Prevents asthma-like symptoms16van der Aa, L.B, et al. Allergy. Published on 2011. 66(2): p. 170-7
Learn more about our specific pre-pro-synbiotics here.
Our unique fermentation process: LactofidusTM
We were also the first to develop a partly fermented infant formula, derived from our unique fermentation process, LactofidusTM.
The process, involving two specific lactic acid-producing bacteria, results in a product containing microbial enzymes, bacterial metabolites such as lactic acid, and bacterial cells and components. Designed to promote digestibility and tolerance, the LactofidusTM fermentation process has been clinically proven to reduce the frequency and intensity of intestinal gas in infants, consequently reducing bloating and GI discomfort.17Roy del. et al. Arch Pediatr. Published on 2004;11: 1546-54
Learn more about the fermentation process in our video.

The new generation of infant formula
We are now bringing together the two pioneering innovations, heralding a new generation of infant nutrition.
The formula will be the first and only that combines our fermented formula (from the Lactofidus™ process) and our patented prebiotic mixture scGOS/lcFOS (9:1).
This innovation has found to be safe and well-tolerated in infants, and enables normal growth;18Huet, et al. J Pediatr Gastroenterol Nutr. Published on 2016; 63:e43-53 importantly, it has also been clinically proven to lower the incidence of colic19Vandenplas, et al. Eur J Pediatr. Published on 2016;321 doi 10.1007/s00431-016-2785-8 and provide softer stools, thereby reducing the incidence of constipation.20Herrera, et al. J Pediatr Gastroenterol Nutr. Published on 2015; 61:516-7 ,21Vandenplas, et al. Arch Dis Child. Published on 2014;99:A91-92
View References
| 1, 8 | Moro, et al. J Pediatr Gastroenterol Nutr. Published on 2002; 34:291- 5 |
|---|---|
| 2, 9 | Boehm, et al. Arch Dis Child. Published on 2002; 86: F178-81 |
| 3 | Knol, et al. J Pediatr Gastroenterol Nutr. Published on 2005a; 40:36-42 |
| 4 | Haarman, et al. Appl Environ Microbiol. Published on 2005; 71:2318-24 |
| 5 | Moro, et al. J Pediatr Gastroenterol Nutr. Published on 2002; 34:291-5 |
| 6 | Knol, et al. J Pediatr Gastroenterol Nutr. Published on 2002; 34:291-5 |
| 7 | Desci, et al. Orv Hetil. Published on 2005; 146:2445-50 |
| 10 | Bruzzese, et al. Clin Nutr. Published on 2009;28:156-161 |
| 11 | Arslanoglu, et al. J Nutr. Published on 2007; 137:2420-2424 |
| 12 | Arslanoglu, et al. J Nutr. Published on 2008; 138:1091-1095 |
| 13 | Arslanoglu, et al. J Biol Regul Homeost Agents. Published on 2012;26:59- 59 |
| 14, 15 | van der Aa, L.B, et al. Clin Exp Allergy. Published on 2010. 40(5): p. 795-804 |
| 16 | van der Aa, L.B, et al. Allergy. Published on 2011. 66(2): p. 170-7 |
| 17 | Roy del. et al. Arch Pediatr. Published on 2004;11: 1546-54 |
| 18 | Huet, et al. J Pediatr Gastroenterol Nutr. Published on 2016; 63:e43-53 |
| 19 | Vandenplas, et al. Eur J Pediatr. Published on 2016;321 doi 10.1007/s00431-016-2785-8 |
| 20 | Herrera, et al. J Pediatr Gastroenterol Nutr. Published on 2015; 61:516-7 |
| 21 | Vandenplas, et al. Arch Dis Child. Published on 2014;99:A91-92 |


